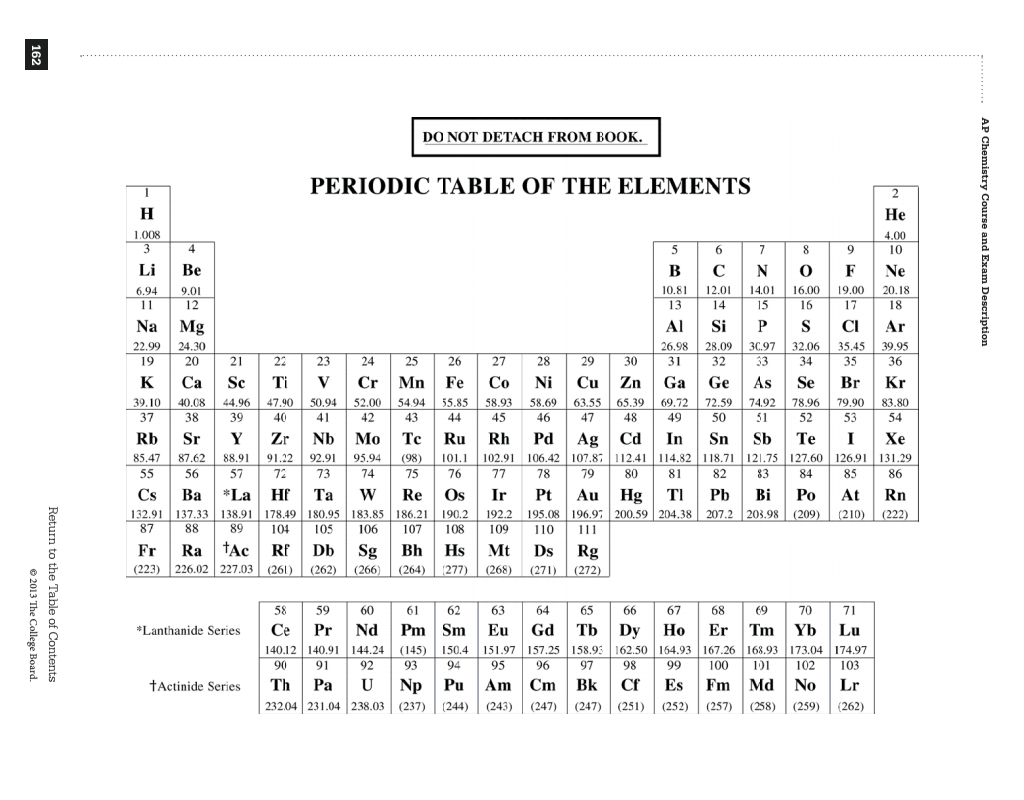
The term 'halogen' means 'salt-former' and compounds that contain one of the halogens are salts. They are located on group 17 of the periodic table and have a charge of -1. Where, generally, are the nonmetals located on the. Where, generally, are the metals located on the periodic table 35. When an atom becomes a cation, what happens to its radius 34. When an atom becomes an anion, what happens to its radius 33. ġ Periodic Table of the Elements Chemistry Reference Sheet California Standards Atomic numberElement symbolAverage atomic mass* Element name Rutherfordium(261)Rf104 11A 22A 1234 77B 111B 122B 133A 166 AKey 8 567 98B 10 144A 155A 177A 188A 33B 44B 55B 66B Technetium(98)Tc43 Polonium(209)Po84 Astatine(210)At85 Pd Radon(222)Rn86 Francium(223)Fr87 Radium(226)Ra88 Actinium(227)Ac89 Dubnium(262)Db105 Seaborgium(266)Sg106 Bohrium(264)Bh107 Hassium(269)Hs108 Meitnerium(268)Mt109 * If this number is in parentheses, then it refers to the atomic mass of the most stable isotope.Ģ Mendelevium(258)Md101 Promethium(145)Pm61 Neptunium(237)Np93 Plutonium(244)Pu94 Americium(243)Am95 Curium(247)Cm96 Berkelium(247)Bk97 Californium(251)Cf98 Einsteinium(252)Es99 Fermium(257)Fm100 Nobelium(259)No102 Lawrencium(262)Lr103 Copyright 2008 California Department of Education Formulas Ideal Gas Law: PV = nRT Calorimetric Formulas P1V1 P2V2 Combined Gas Law: = No Phase Change: Q = m( T)CpT1 T2 Pressure Formula: P= F Latent Heat of Fusion: Q = m HfusA Mass-Energy Formula: E = mc2 Latent Heat of Vaporization: Q = m Hvap Constants LVolume of Ideal Gas at STP: mol Speed of Light in a Vacuum: c = 108 ms Specific Heat of Water: Cp(H2O) = cal J (gC)= (gC) cal JLatent Heat of Fusion of Water: Hfus(H2O)= 80 g = 334 g cal JLatent Heat of Vaporization of Water: Hvap(H2O)= 540 g = 2260 g Unit Conversions Calorie-Joule Conversion: 1 cal = J Absolute Temperature Conversion: K= C + 273 Conversions: 1 atm = 760 mm Hg = 760 Torr = kPa = in. Halogens are comprised of the five nonmetal elements Flourine, Chlorine, Bromine, Iodine, and Astatine. What is the general trend of ionization energy as you go down a group on the periodic table 32. 
Periodic Table of the Elements Chemistry Reference Sheet California Standards Test Sodium 22.99 Na 11 Atomic number Element symbol Average atomic mass* Element name Hydrogen 1.01 H 1 Lithium 6.94 Li 3 Sodium 22.99 Na 11 Potassium 39.10 19 K Nickel 58.69 Ni 28 Rubidium 85.47 Rb 37 Rutherfordium (261) Rf 104 Molybdenum 95.94 Mo 42 Germanium 72.61. Periodic Table of the Elements Chemistry Reference Sheet California Standards Atomic numberElement symbolAverage atomic mass* Element name Rutherfordium(261)Rf104 11A 22A 1234 77B 111B 122B 133A 166 AKey 8 567 98B 10 144A 155A 177A 188A 33B 44B 55B 66B Technetium(98)Tc43 Polonium(209)Po84 Astatine(210)At85 Pd Radon(222)Rn86 Francium(223)Fr87 Radium(226)Ra88 Actinium(227)Ac89 Dubnium(262)Db105 Seaborgium(266)Sg106 Bohrium(264)Bh107 Hassium(269)Hs108 Meitnerium(268)Mt109 * If this number is in parentheses, then it refers to the atomic mass of the most stable isotope. 0 ratings 0 found this document useful (0 votes) 326 views. Chemistry Reference Sheet: Periodic Table of The Elements California Standards Test. Example: marketing Search Periodic Table of the Elements Chemistry Reference Sheet. Chem Toe - Free download as PDF File (.pdf), Text File (.txt) or view presentation slides online.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
